Introduction
PD-1,or ‘programmed-death 1’, was initially considered to be a molecule that regulates cell death, but is now identified as a key immune checkpoint inhibitory receptor, which is expressed on activated tumour-specific CD4+ helper and CD8+ killer T lymphocytes. Therapeutic antibody-mediated blockade of PD-1 or one of its ligands, PD-L1, removes the suppressive effects of PD-L1 on cytotoxic T-cells with restoration of host immunity against the tumour. For some patients, this treatment approach can control or eliminate advanced malignant neoplasms, including metastatic melanoma 1, which has fostered intense research interest 2.
Definition of PD-1/PD-L1
PD-1 (CD279) is a cell surface receptor that is part of the immunoglobulin superfamily that is expressed primarily on the surface of activated T-cells 3,4. PD-1 was initially considered to be a molecule that regulates cell death but is now identified as a key immune checkpoint inhibitory receptor. PD-L1 or ‘programmed-death ligand 1’ (CD274), the main PD-1 ligand, is a transmembrane protein expressed on a variety of cell types, including antigen presenting cells, mainly dendritic cells and macrophages 3 and constitutively expressed by non-lymphoid tissues including heart, lung and others 5. Binding of PD-L1 inhibits the function of activated T-cells, which is an important mechanism for negative feedback control of inflammation and autoimmunity in the peripheral effector phase of T-cell activation 3 and identifies the PD-1/PD-L1 pathway as a significant immune response checkpoint.
Tumour cells can co-opt this PD-1/PD-L1 regulatory mechanism. Tumour cells may express PD-L1, with subsequent PD-1 binding and inhibition of T-cell activation allowing cancer cells to evade immune attack. PD-L1 expression can be driven by both adaptive immune resistance (through IFN-g receptor signalling) as well as oncogenic mechanisms (e.g. gene amplification) 6. A range of solid and haematological malignancies over-express PD-L1 3. Pre-clinical studies that found decreased tumour growth and improved survival with PD-1/PD-L1 pathway blockade provided the rationale for immune checkpoint inhibition as a novel approach to cancer treatment.
Drugs Targeting the PD-1/PD-L1 Pathway
A small, first-in-human trial of the anti-PD-1 drug nivolumab reported promising results, and subsequently, a large trial in five different cancer types found durable tumour regressions in a substantial proportion of otherwise refractory melanomas, kidney cancers, and lung cancers 1,2. Blockade of the PD-1/PD-L1 pathway has now emerged from being a promising cancer therapy to an established class of drugs that are indicated across multiple cancer types 2.
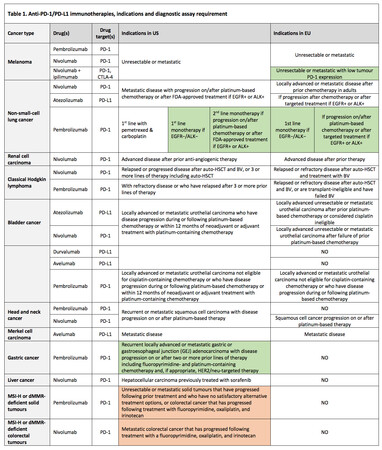
Currently, five monoclonal antibodies (two PD-1 inhibitors and three PD-L1 inhibitors) have been developed and licensed for clinical use (see Table 1), and many more are in pre-clinical and clinical development. These immunotherapies can disrupt the engagement of PD-1 with its ligands and impede inhibitory signals in T-cells, with resultant tumour recognition by cytotoxic T-cells. Remarkable clinical responses have been seen in some patients in various cancer types, including melanoma, lung, kidney, bladder cancer and others. Several hundreds of ongoing clinical trials are investigating immune checkpoint blockade therapies both alone and in combination, as well as with vaccines, metabolic drugs, chemotherapy and kinase inhibitors to treat a variety of cancers 2,6.
Anti-PD-1 drugs
Two PD-1 inhibitors are approved for clinical use in both Europe and the US—pembrolizumab (Keytruda®) and nivolumab (Opdivo®) (see Table 1). Both are human IgG4 monoclonal antibodies that block PD-1 and are indicated for the treatment of a broad range of tumour types 7-10.
Anti-PD-L1 drugs
Three PD-L1 inhibitors—atezolizumab (Tecentriq®), avelumab (Bavencio®), and durvalumab (Imfinzi®)—are approved cancer immunotherapies for several advanced cancers 11- 15, atezolizumab and avelumab in both Europe and US, and durvalumab in the US only (although the European Medicines Agency (EMA) has recently accepted a marketing authorisation application for durvalumab) (Table 1).
PD-L1 as a Predictive Biomarker
Defining biomarkers that predict therapeutic response to PD-1/PD-L1 blockade is an important goal 16. The first study that provided evidence of a link between PD-L1 protein expression by tumour cells detected by immunohistochemistry (IHC) and response to anti-PD-1 therapy was the first-in-human phase I nivolumab trial conducted in patients with a variety of different solid tumour types 17.
Subsequently, a number of trials validated that PD-L1 expression correlates with an increased response to PD-1 and PD-L1 immune checkpoint inhibitors, but the response rate was lower than the early studies.
Examples of PD-L1 IHC are shown in Figure 1. Patients without PD-L1 expression can also derive benefit from these agents, with studies across multiple cancer types demonstrating a pooled response rate of 48% in patients with PD-L1-positive tumours compared with 15% in PD-L1-negative tumours 18. Thus, while PD-L1 expression has been associated with more favourable response rates to PD-1/PD-L1 checkpoint inhibitors, PD-L1 does not appear to be a static biomarker and does not offer binary discrimination of responsiveness.
In melanoma, non–small cell lung carcinoma (NSCLC), and bladder cancer, PD-L1 IHC has identified patients with a higher likelihood of treatment response to certain drugs. In the US, there have been four evidence-based FDA approvals of commercial IHC tests linked to specific PD-1/PD-L1 pathway drugs and cancer types 19-22. Only pembrolizumab has indications restricted to tumours expressing PD-L1 (NSCLC and gastric cancer) and requires the use of a companion diagnostic. Currently, PD-L1 IHC 22C3 pharmDx (Dako) is the only FDA-approved companion diagnostic, which is used to select patients for treatment with pembrolizumab. The other three FDA-approved PD-L1 IHC assays are a complementary test that may provide physicians more information and inform patient dialogue when deciding treatment. These are PD-L1 IHC 28-8 pharmDx assay for nivolumab treatment, VENTANA PD-L1 IHC (SP142) assay for atezolizumab treatment and VENTANA PD-L1 IHC (SP263) assay for durvalumab. The wording of the licences has distinct recommendations regarding PD-L1 expression that reflect the clinical trial evidence in different tumour types.
Pembrolizumab
The approval of pembrolizumab in NSCLC was based on results of two randomised, controlled trials that demonstrated statistically significant improvements in progression-free survival (PFS) and overall survival (OS) for patients randomised to pembrolizumab compared with chemotherapy. In the KEYNOTE-024 trial of 305 patients with metastatic NSCLC, whose tumours had a high PD-L1 expression (expressed in ≥ 50% tumour cells), had not received prior chemotherapy and did not have EGFR or ALK genomic tumour aberrations, those who received pembrolizumab had a significant improvement in PFS and OS versus chemotherapy: OS (hazard ratio [HR] 0.60; 95% confidence interval [CI]: 0.41–0.89; p=0.005), and PFS (HR 0.50; 95% CI: 0.37–0.68; p<0.001) 23. In the KEYNOTE-010 trial of 1033 patients who were previously treated for metastatic NSCLC and expressed PD-L1 in ≥1% tumour cells, those randomised to pembrolizumab had an improved OS compared with patients receiving docetaxel. The HR for OS was 0.71 (95% CI: 0.58–0.88), p<0.001 comparing pembrolizumab 2 mg/kg with chemotherapy, and OS was 0.61 (95% CI: 0.49–0.75), p<0.001 comparing pembrolizumab 10 mg/kg with chemotherapy 24.
In KEYNOTE-059, cohort 1 included 259 patients with advanced gastric or gastroesophageal junction cancer who had progressed on at least two prior lines of chemotherapy who received pembrolizumab 25. After a median follow-up of 6 months, patients who expressed PD-L1 (combined positive score (CPS) ≥ 1) were more likely to respond than those who did not, with objective response rates (ORR) of 16% and 6%, respectively. This is reflected in the wording of the indication, restricting use to tumours that express PD-L1 (CPS≥ 1) 9. The FDA recommends assessing the feasibility of a fresh tumour biopsy if PD-L1 expression is not detected in an archival specimen.
Nivolumab
The EMA and FDA licensed both pembrolizumab and nivolumab for treatment of advanced melanoma independent of PD-L1 expression. The combination of nivolumab with ipilimumab in advanced melanoma improved PFS over nivolumab alone, in patients with a low tumour PD-L1 expression measured by the PD-L1 IHC 28-8 pharmDx assay 26, and this finding is reflected in the wording of the nivolumab melanoma indication in the EU 7.
PD-L1 positivity is also associated with improved survival from nivolumab treatment in non-squamous NSCLC 27. The predictive value of PD-L1, using the PD-L1 IHC 28-8 pharmDx assay, was seen across all the efficacy endpoints of the CheckMate 057 study at an expression level ≥1% 27. Despite these findings, a companion diagnostic is not required for nivolumab treatment in melanoma or NSCLC 7,8. This decision was likely influenced by limitations of the clinical data to support the IHC assay, for example, findings from clinical trials that demonstrated similar outcomes irrespective of PD-L1 expression status 28,29. Furthermore, testing for PD-L1 expression in the CheckMate 057 study was retrospective and was not performed on all patient specimens but rather a subset of 78% of cases, with the potential risk of selection bias 28.
Atezolizumab
In a non-randomised, prospective, single-arm study of atezolizumab, PD-L1 expression in ≥ 5% tumour-infiltrating immune cells, determined by the VENTANA PD-L1 (SP142) Assay, in urothelial carcinoma tissue was associated with increased ORR 30. In patients who were classified as positive for PD-L1 expression (≥ 5% tumour-infiltrating immune cells), 26% of participants experienced a tumour response compared to 9.5% of participants who were classified as negative for PD-L1 expression (< 5% tumour-infiltrating immune cells). Therefore, patients experienced a tumour response across the study; however, the greater effect in those who were classified as positive for PD-L1 expression suggests that the level of PD-L1 expression in tumour infiltrating immune cells may help identify patients whose tumours are more likely to respond 21. Hence, the VENTANA PD-L1 (SP142) Assay has been approved by the FDA as a complementary diagnostic to help physicians decide which patients with urothelial cancer are most likely to respond to atezolizumab 21.
Durvalumab
Durvalumab’s application received accelerated approval, priority review, and Breakthrough Therapy Designation from the FDA, based on one single-arm phase I/II trial of locally advanced or metastatic urothelial carcinoma patients whose disease progressed after prior platinum-containing chemotherapy. PD-L1 expression on tumour cells and infiltrating immune cells (IC) was determined prospectively using the VENTANA PD-L1 (SP263) Assay. This Assay has been approved by the FDA, as a complementary diagnostic for assessing PD-L1 in urothelial carcinoma tissue 22. Of 182 patients, 95 were PD-L1 high (defined as TC and IC ≥25% PD-L1 positive if IC was >1% of the tumour area or TC ≥25% and IC=100% positive if IC was ≤1%) and 73 were PD-L1 low 15. Overall, the ORR was 17.0%; when analysed by PD-L1 expression status, as measured by the approved assay, ORR was 26.3% in high PD-L1 score and 4.1% in low PD-L1 score 15.
Quality Testing Results
The conflicting observations regarding PD-L1 as a predictive biomarker of tumour response likely reflects a number of issues, both IHC-test specific and tumour-biology-related. These include the limitations inherent in tumour sampling, with focal expression potentially missed in small biopsies, and dynamic PD-L1 expression apparent over time and by anatomical site in individual patients. Different IHC detection methods and antibodies, quality of samples, methods used to acquire material, and positivity thresholds may also be a factor 6.
Recognising these issues, a Blueprint Working Group has been established with cooperation from the pharmaceutical industry to provide a comparison of different IHC tests and cell scoring methods for PD-L1 expression, reflecting that each drug is developed in the context of a unique biological scientific hypothesis and registration strategy. The initial findings show that the percentage of PD-L1-stained tumour cells was comparable when the 22C3, 28-8, and SP263 assays were used, whereas the SP142 assay exhibited fewer stained tumour cells. By comparing assays and cut-offs, the Group concluded that more data are required before an alternative assay can be used to read different specific therapy-related PD-L1 cut-offs 31. Regarding the tumour microenvironment, PD-L1 can be expressed across a wide range of cell types, creating challenges in individual cell identification 4. The impact on the tumour microenvironment that may have occurred through multiple previous cancer treatments remains undefined 6.
Patient Selection
As a result, and highlighted here, PD-L1 IHC positivity is an imperfect biomarker of response and currently not suitable as a definite biomarker for selection for therapy with PD-1/PD-L1 inhibitors 6,18. It is likely that a more complex, multicomponent predictive biomarker system will be required to refine appropriate patient selection for PD-1/PD-L1 blockade 6,32.
References
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 2012;366:2443-54.
- Topalian SL. Targeting immune checkpoints in cancer therapy. JAMA 2017;318(17):1647-1648. doi:10.1001/jama.2017.14155.
- Patel SP, Kurzrock R. PD-L1 Expression as a Predictive Biomarker in Cancer Immunotherapy. Mol Cancer Ther 2015;14:847-56.
- Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus docetaxel in advanced squamous-cell non–small-cell lung cancer. N Engl J Med 2015;373:123-35.
- Suarez ER, Chang de K, Sun J, et al. Chimeric antigen receptor T cells secreting anti-PD-L1 antibodies more effectively regress renal cell carcinoma in a humanized mouse model. Oncotarget 2016;7(23):34341-55.
- Topalian SL, Taube JM, Anders RA, et al. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer 2016;5:275-87.
- Opdivo® Summary of Product Characteristics, 2017.
- Opdivo® prescribing information, 2017.
- Keytruda® prescribing information, 2017.
- Keytruda® Summary of Product Characteristics, 2017.
- Tecentriq® Prescribing Information, 2017.
- Tecentriq® Summary of Product Characteristics, 2017.
- Bavencio® Prescribing Information, 2017.
- Bavencio® Summary of Product Characteristics, 2017.
- Imfinzi® Prescribing Information, 2017.
- Bhaijee F, Anders RA. PD-L1 Expression as a Predictive Biomarker: Is Absence of Proof the Same as Proof of Absence? JAMA Oncol 2016;2(1):54-5.
- Brahmer JR, Drake CG, Wollner I, et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol 2010;28:3167-75.
- Sunshine J, Taube JM. PD-1/PD-L1 inhibitors. Curr Opin Pharmacol 2015;23:32-8.
- PD-L1 IHC 22C3 pharmDx FDA Summary of Safety and Effectiveness Data, 2017.
- PD-L1 IHC 28-8 pharmDx FDA Summary of Safety and Effectiveness Data, 2016.
- VENTANA PD-L1 (SP142) Assay FDA Summary of Safety and Effectiveness Data, 2016.
- VENTANA PD-L1 (SP263) Assay FDA Summary of Safety and Effectiveness Data, 2017.
- Reck M, Rodrıguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016;375:1823–33.
- Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 2016;387:1540–50.
- Wainberg ZA, Jalal S, Muro K, et al. KEYNOTE-059 Update: Efficacy and safety of pembrolizumab alone or in combination with chemotherapy in patients with advanced gastric or gastroesophageal (G/GEJ) cancer. Abstract LBA28_PR. ESMO Congress 2017.
- Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilumab or monotherapy in untreated melanoma. N Engl J Med 2015;373:23-34.
- Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced non-squamous non-small cell lung cancer. N Engl JMed 2015;373:1627–1639.
- Jørgensen JT. Companion diagnostic assays for PD-1/PD-L1 checkpoint inhibitors in NSCLC. Expert Rev Mol Diagn 2016;16:131-3.
- Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320–30.
- Rosenberg JE, Hoffman-Censits J, Powles T, et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 2016;387:1909-20.
- Hirsch FR, McElhinny A, Stanforth D, et al. PD-L1 Immunohistochemistry Assays for Lung Cancer: Results from Phase 1 of the Blueprint PD-L1 IHC Assay Comparison Project. J Thorac Oncol 2017;12(2):208-22.
- Boussiotis VA. Molecular and Biochemical Aspects of the PD-1 Checkpoint Pathway. N Engl J Med 2016;375:1767-78.